Inflammatory bowel disease (IBD) is actually an immune-mediated chronic intestinal condition. It is more commonly found in women during adolescence or in young adults. 1 There are several factors which are predicted to be the causes of IBD, such as genetics, mucosal immune responses, epithelial defects, and combination of errant host interactions with intestinal microbiota. 1-2
Pathogenesis of Inflammatory Bowel Disease (IBD)
Genetics
IBD is a hereditary disease that carries a 10% possibility of first-degree relative being affected. Children with IBD affected parents may have a 36% chance of being affected. Moreover, it is also stated that Ulcerative Colitis and Crohn’s Disease (major types of IBD) are associated with Turner’s syndrome, while Hermansky-Pudlak syndrome is associated with granulomatous colitis, another type of IBD. The table below explains the differences of tendencies leading to UC and CD. Furthermore, the occurrence of CD is often correlated with NOD2 polymorphisms in which certain variants of NOD2 polymorphisms are not effective enough in combating luminal microbes which are why they are able to enter lamina propria and trigger inflammatory reactions. 1-2

Tabel 1. Epidemiology of IBD Harrison’s Gastroenterology and Hepatology. 2010.
Mucosal immune responses
Polarization of helper T cells to the TH1 type and TH17T cells are suggested be the factors contributing to the occurrence of the disease. Moreover, certain polymorphisms from IL-23 also gives protection from CD and UC while mucosal IL-13 production may increase in UC (lesser in CD), pointing that combination of mucosal immunity activation and immunoregulation suppression contribute to the development of UC and CD. 1
Epithelial defects
Certain NOD2 polymorphisms may be associated with the epithelial integrity, regarding the defects in intestinal epithelial tight junction barrier. However, it is also stated that CD patients with ATGI6LI mutations have abnormal Paneth cell granules which supposedly carry antimicrobial peptides. 1
Microbiota
As we know, the quantity of microbial organisms in the gastrointestinal lumen is large. The precise role of microbiota to IBD pathogenesis remains unclear despite of growing data of their effects to IBD pathogenesis. 1
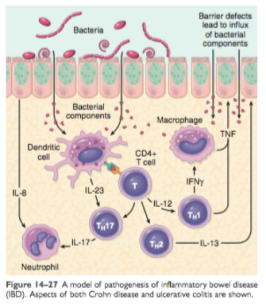
Figure 1. Microscopic view of Ulcerative Colitis Robbins Basic Pathology 9th Ed. 2013.
When IBD is initiated, the immune inflammatory response is perpetuated by T-cell activation; activating such cytokines as IL-1, IL-6, and TNF to promote fibrogenesis, collagen production, activation of tissue metalloproteinases, and activation of blood coagulation factor in localized tissue. These activations of proinflammatory cells are not in balance with anti-inflammatory mediators, causing prolonged inflammation of the bowel. 1
There are 2 (two) major types of Inflammatory Bowel Disease (IBD), which are Ulcerative Colitis and Crohn’s Disease.
Ulcerative Colitis
Ulceratic Colitis (UC) is a mucosal disease which is often founded in rectum and extends towards proximal, involving all or part of the colon. It is an inflammatory reaction of the colon in which there is erythematous mucosa with a fine granular surface which looks like sandpaper in mild inflammation case. In severe inflammation, the mucosa is hemorrhagic, edematous, and ulcerated. In long-standing (chronic) disease, there might be inflammatory polyps (pseudopolyps) as a result of epithelial regeneration. The mucosa appears normal at first, however it proceeds to appear atrophic and featureless; thus, making the whole colon appears narrowed and shortened. 2
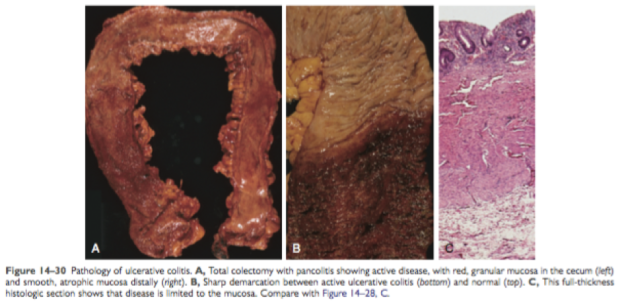
Figure 2. Macroscopic view of Ulcerative Colitis Robbins Basic Pathology 9th Ed. 2013.
Microscopically, we can see a distorted crypt architecture of the colon and the crypts may be bifid and reduced in number. Multiple basal lymphoid and basal plasma cells aggregates may also be seen. Additionally, neutrophils invasion can also be seen in the crypts, giving rise to cryptitis and crypt abscesses. 2
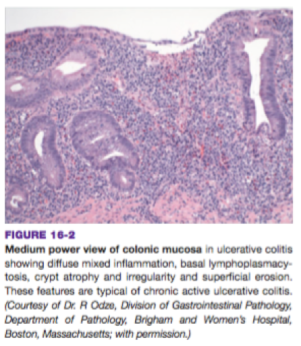
Figure 3. Microscopic view of Ulcerative Colitis Harrison’s Gastroenterology and Hepatology. 2010.
There are several sites of predilection in UC and we can classify them based on the locations. For example, pancolitis is UC which occurs at the entire colon. Disease limited to the rectum or rectosigmoid will be named as ulcerative proctitis or ulcerative proctosigmoiditis. Moreover, if it is located in the distal ileum, it will be called backwash ileitis. 1
Unlike in CD, there is no mural thickening, serosal surface appears normal and there is hardly any strictures occur. Nevertheless, the inflammation can damage muscularis propria and disturb neuromuscular function which may lead to toxic megacolon, carries high risk of perforation. Moreover, granulomas are also not present in UC. 1
UC has the symptoms of having diarrhea, rectal bleeding, tenesmus, passage of mucus, and crampy abdominal pain. Colonic motility is also altered by inflammation with rapid transit through the inflamed intestine. However, the diarrhea is mostly nocturnal. 2
Crohn’s Disease
Different with UC, Chron’s Disease (CD) can affect the whole gastrointestinal tract, starting from mouth to anus. However, the rectum area is commonly spared in CD condition (unlike CD). However, CD inflammatory lesions are segmental with skip areas in the midst of diseased intestine. 2
Small superficial ulcerations characterize mild disease while stellate ulcerations fuse longitudinally and transversely in the more active disease. However, pseudopolyps can also be found in CD similarly to UC. However, the CD lesions are transmural while UC lesions are only limited to mucosa and submucosa layers. Active CD is characterized by focal inflammation and formation of fistula tracts, which resolve by fibrosis and structuring of the bowel. Bowel wall thickens and becomes narrowed and fibrotic, proceeds to chronic recurrent bowel obstruction. 2 In severe case of transmural disease, mesenteric fat frequently extends around the serosal surface, this condition is commonly called as creeping fat. 1
Crypt abscess, groups of neutrophils infiltration in colonic crypt, will destruct the integrity of the colon These destructions will be followed by regeneration phase. This cycle will go on and lead to distortion of mucosal architecture, making the colonic crypts form bizarre branching shapes and unusual orientations to one another (which normally should be straight and parallel) 1
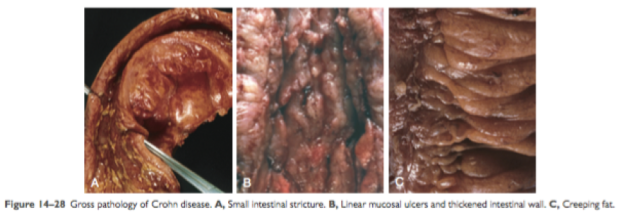
Figure 5. Macroscopic view of Crohn’s Disease. Robbins Basic Pathology 9th Ed. 2013.
As is explained before, aphthoid ulcerations and focal crypt abscesses with loose aggregations of macrophages form noncaseating granulomas in all of the bowel wall. Granulomas can be seen in lymph nodes, mesentery, peritoneum, liver, and pancreas. Furthermore, thickened mesentery projections which encase the colon, together with serosal and mesenteric inflammation will promote adhesions and fistula formation. 1-2
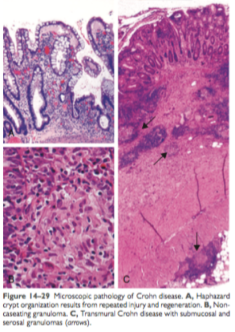
Figure 6. Microscopic view of Crohn’s Disease. Robbins Basic Pathology 9th Ed. 2013.
Moreover, there is also tendency of CD to lead to epithelial metaplasia which often takes the form of gastric antral-appearing glands (pseudopyloric metaplasia). Moreover, Paneth cell metaplasia may also occur in the left colon. Not to mention the noncaseating franylomas, which may rise in any area of intestinal wall (in both involved/diseased or uninvolved regions). 1
Patients with CD often have intermittent attacks of mild bloody diarrhea, fever, abdominal pain in lower right quadrant. This disease can be reactivated (thus resulting in recurrent symptoms) due to such external stimulations as physical or emotional stress, cigarette smoking, and specific dietary items. 1
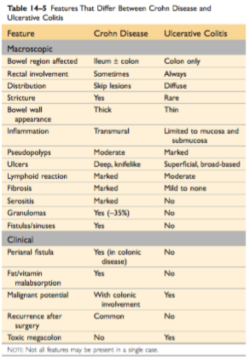
Tabel 2. Differentiating CD and UC. Robbins Basic Pathology 9th Ed. 2013.
Indeterminate Colitis
UC and CD have similar symptoms and clinical manifestations to other diseases. For differentiating UC and CD themselves, they are often hard to be distinguished in up to 15% cases. This condition is called indeterminate colitis. 2 Patients with overlapping clinical symptoms of certain patients might still be treated effectively. However, it is recommended to differentiate UC and CD while we can as the evolving medical therapies and surgical management are specific to each type of IBD. 1 We can differentiate CD and UC by several aspects which have already been shown in the previous table and the table below:
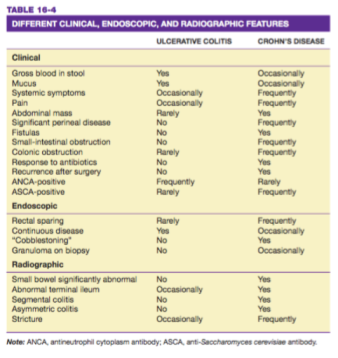
Tabel 3. CD and UC differences based on clinical, endoscopic, and radiographic features. Harrison’s Gastroenterology and Hepatology. 2010.
References
- Kumar V, Abbas AK, Aster JC. Robbins Basic Pathology 9th Philadelphia: Elsevier; 2013: 587-91.
- Longo DL, Fauci AS. Harrison’s Gastroenterology and Hepatology. NewYork: The McGraw-Hill; 2010: 174-83.
